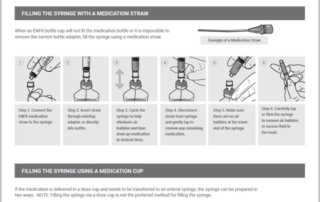
ENFit Implementation: Forming a Team, Building & Executing the Plan
Date: Mar 16, 2017 Length:4:52
ENFit Implementation: Pharmacy Keys to Success
Date: March 16th, 2017 | Length 3:56
ENFit Implementation: Materials Distribution Keys to Success
https://www.youtube.com/watch?v=BEy79YOdB0Q
ENFit Implementation: Clinical Education Methods & Tools
https://www.youtube.com/watch?v=cJ0PJh3f_h4
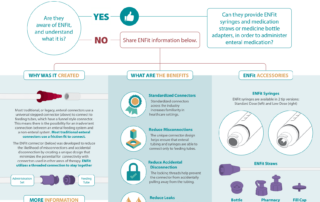
NRFit / NHS Flyer
Link to the Full NHS Alert Here
LATAM Flyer – Portuguese
Mais recursos portugueses
LATAM Flyer – Spanish
Más recursos en español
ISO Series Flyer
ENFit and NRFit represent enteral and neuraxial patient safety initiatives, but they are just 2 of the 6 sections of the ISO 80369 series of standards.
Pediatric Health Alliance – GEDSA – ENFit & NRFit Webinar
https://www.youtube.com/watch?v=Z8gKx5HljjA
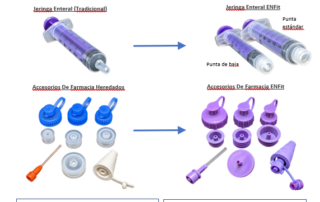
ENFit Pharmacy Resource Guide – Spanish
Resources for Providers Resources for
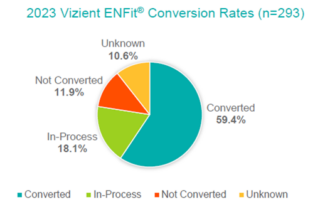
GEDSA US Conversion Rates Flyer 2023
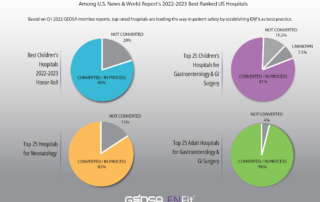
Top ranked US hospitals are leading the way in establishing ENFit as a best practice for patient safety.
ENFit Dietitian Cleveland Clinic & GEDSA Webinar
https://www.youtube.com/watch?v=IX3sGqxfdI8