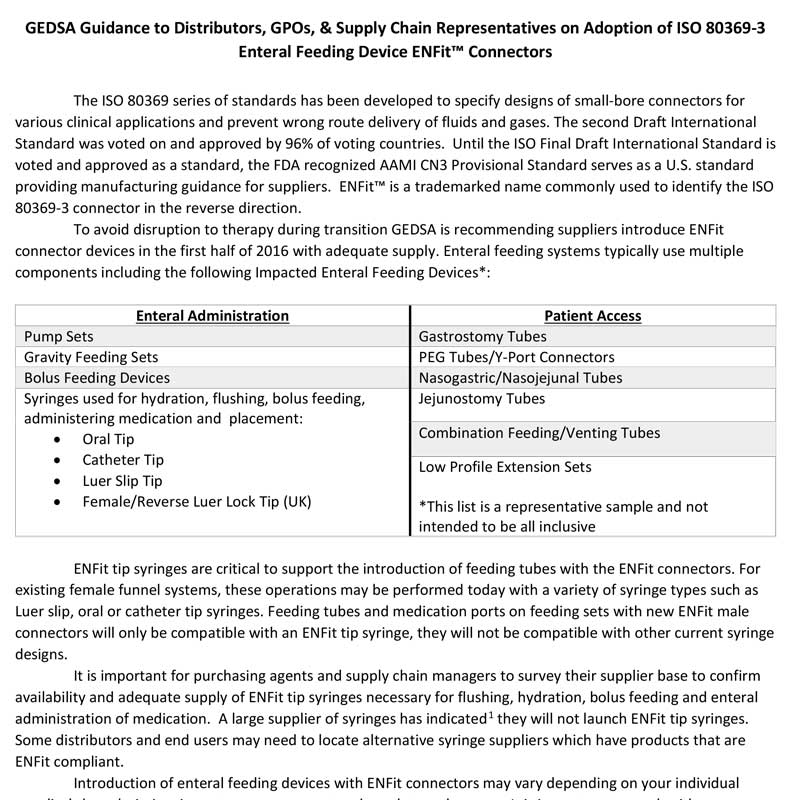
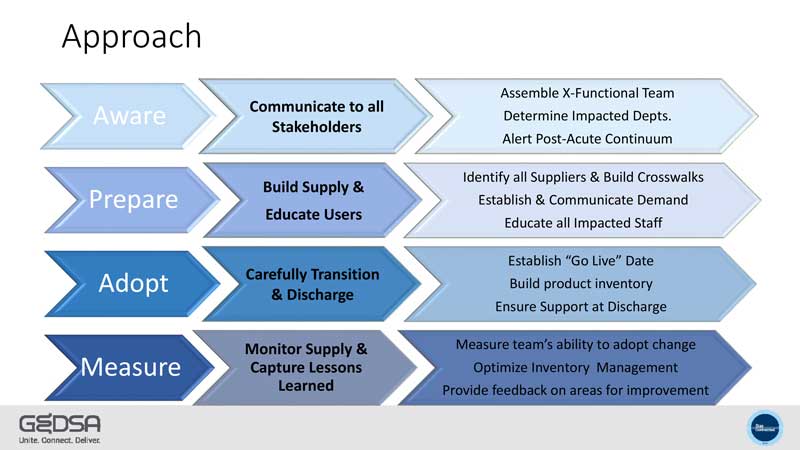
GEDSA Guidance to Distributors, GPOs, & Supply Chain Representatives on Adoption of ISO 80369-3 Enteral Feeding Device ENFit™ Connectors
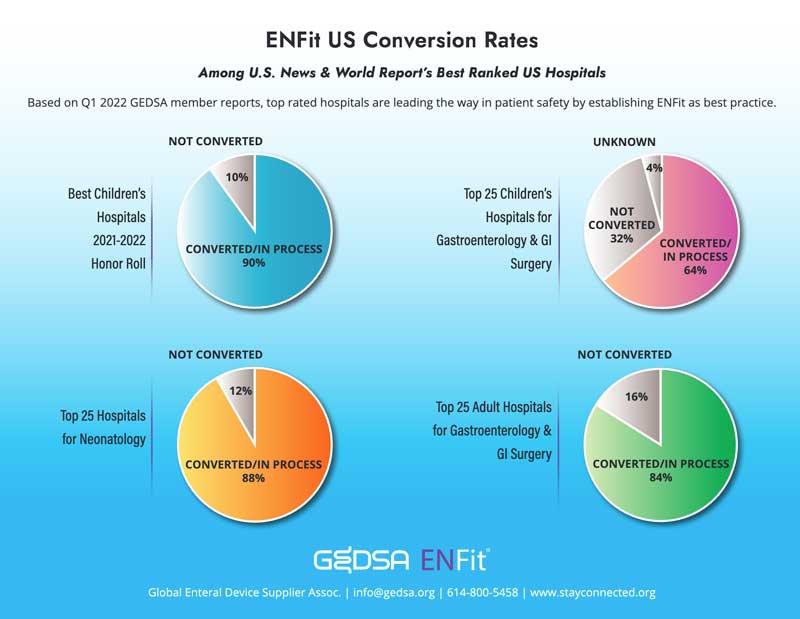
Subject Line: The FDA Encourages Use of Enteral Device Connectors that Reduce Risk of Misconnection and Patient Injury Dear Colleagues, The U.S. Food and Drug Administration (FDA) is concerned by continued reports of misconnections with enteral devices. To reduce the risk of misconnections and patient injury, the FDA recommends hospitals and clinicians use enteral devices with connectors that meet the International Organization for Standardization (ISO) 80369-1 or ISO 80369-3 standard, or that are otherwise designed to reduce the risk of misconnections. There are currently marketed enteral connectors that meet the 80369-3 standards, many of which are identified by the tradename ENFit.
This paper shows the method for using table 1 in ISO 7886-1 to calculate the Tolerance on graduated capacity (TOGC) for syringes, and displays calculated values for different combinations of syringe size (1, 5 and 10ml) and expelled volume. It is available online as a pdf.
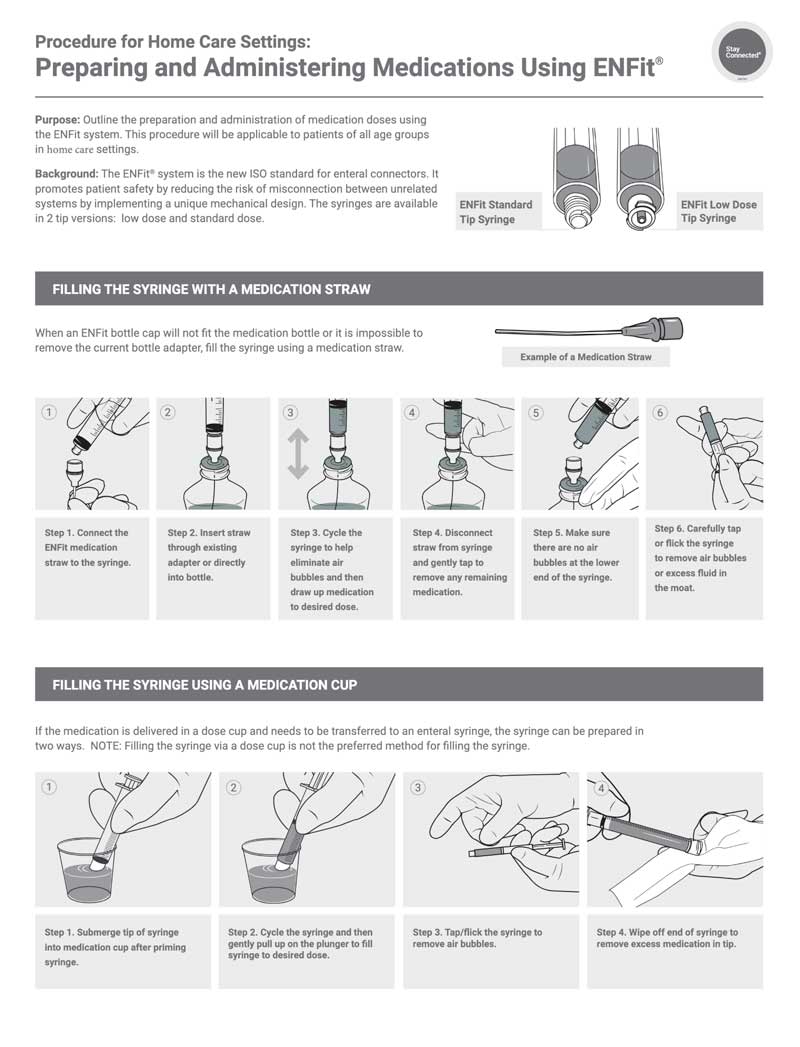
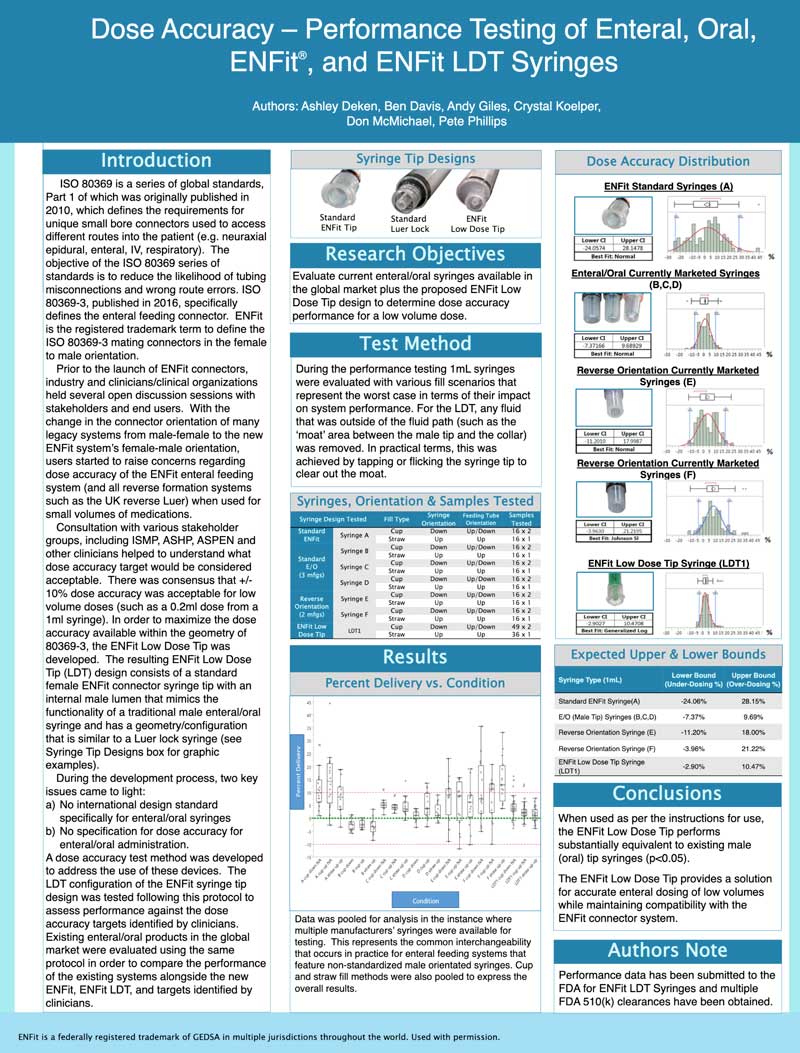
Purpose: Outline the preparation and administration of medication doses using the ENFit system. This procedure will be applicable to patients of all age groups in inpatient settings. Background: The ENFit® system is the new ISO standard for enteral connectors. It promotes patient safety by reducing the risk of misconnection between unrelated systems by implementing a unique mechanical design. The syringes are available in 2 tip versions: low dose and standard dose.
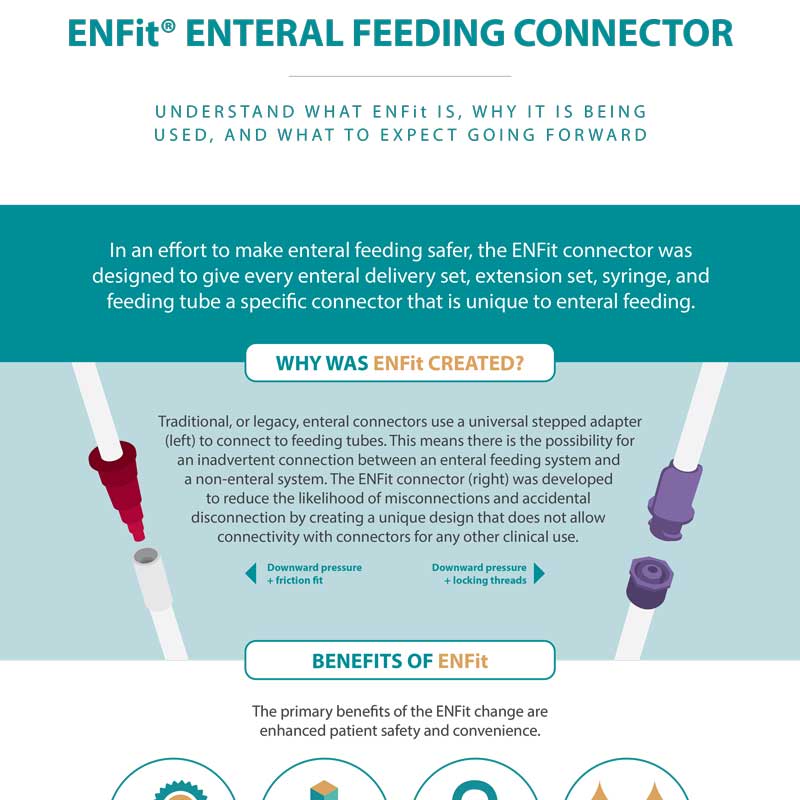
In an effort to make enteral feeding safer, the ENFit connector was designed to give every enteral delivery set, extension set, syringe, and feeding tube a specific connector that is unique to enteral feeding.